Learn More

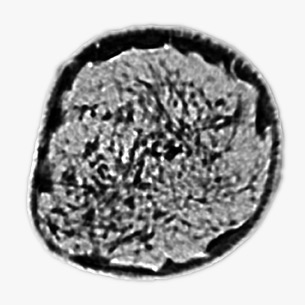
Dense breasts are common. In fact, about 50% of women in the US and more than 75% of women in Asia have dense breasts. With images that show clear information about breast tissue, our goal is to identify what is benign, and what’s problematic.
See for yourself* Source: Mark Lenox et al. Imaging Performance of Quantitative Transmission Ultrasound, International Journal of Biomedical Imaging
1 Source: 3-D Nonlinear Acoustic Inverse Scattering: Algorithm and Quantitative Results, IEEE Transactions on Ultrasonics, Ferroelectrics, and Frequency Control
2 Source: Imaging Performance of Quantitative Transmission Ultrasound, International Journal of Biomedical Imaging
3 Source: Breast Biopsy: Procedure Types, What to Expect and Results, National Breast Cancer Foundation
4 Source: U.S. Breast Cancer Statistics, Breastcancer.org
B. Malik, R. Terry, J. Wiskin, and M. Lenox, Quantitative Transmission Ultrasound Tomography: Imaging and Performance Characteristics, Medical Physics, published 2019.